 Prioritize...
Prioritize...
After completing this section, you should be able to recite and explain the four laws of radiation. Your explanations should contain specific examples because you will be required to apply and integrate these laws in your understanding of atmospheric remote sensing. Read...
Read...
In order to best make use of the of information that comes to us via the electromagnetic spectrum, we need to understand some basic properties of radiation. A complete treatment on the subject of radiation theory would take an entire course at least (indeed, folks pursuing a BS in meteorology are usually required to take a Radiative Transfer course). Instead, you just need to know the fundamental principles describing the electromagnetic radiation that originates from an object, and how that radiation travels through space (discussed in the next section).
For electromagnetic radiation, there are four "laws" that describe the type and amount of energy being emitted by an object. In science, a law is used to describe a body of observations. At the time the law is established, no exceptions have been found that contradict it. The difference between a law and a theory is that a law simply describes something, while a theory tries to explain "why" something occurs. As you read through the laws below, think about observations from everyday life that you have made that might support the existence of each law.
Planck's Law
Planck's Law can be generalized as such: Every object emits radiation at all times and at all wavelengths. If you think about it, this law is pretty hard to wrap your brain around. We know that the sun emits visible light (below left), infrared waves, and ultraviolet waves (below right), but did you know that the sun also emits microwaves, radio waves, and X-rays? OK... you are probably saying, the sun is a big nuclear furnace, so it makes sense that it emits all sorts of electromagnetic radiation. However, Plank's Law states that every object emits over the entire electromagnetic spectrum. That means that you emit radiation at all wavelengths -- so does everything around you!

Now before you dismiss this statement out-of-hand, let me say that you are not emitting X-rays in any measurable amount (thank goodness!). The mathematics behind Plank's Law hinge on the fact that there is a wide distribution of vibration speeds for the molecules in a substance. This means that it is possible for matter to emit radiation at any wavelength, and in fact it does.
Another common misconception that Plank's Law dispels is that matter selectively emits radiation. Consider what happens when you turn off a light bulb. Is it still emitting radiation? You might be tempted to say "No" because the light is off. However, Plank's Law tells us that while the light bulb may no longer be emitting radiation that we can see, it is still emitting at all wavelengths (most likely, it is emitting copious amounts of infrared radiation). Another example that you hear occasionally on TV weathercasts goes something like this. "When the sun sets, the ground begins to emit infrared radiation..." This is certainly not true by nature of Planck's Law (and besides, how does the ground know when the sun sets anyway). We'll talk more about radiation emission from the ground in a future lesson. For now, please dismiss such statements as hogwash. The surface of the earth emits radiation all the time and at all wavelengths.
Wein's Law
At this point I know what you are thinking... there must be a "catch". In fact there is. While all matter emits radiation at all wavelengths, it does not do so equally. This is where the next radiation law comes in. Wein's Law states that the wavelength of peak emission is inversely proportional to the temperature of the emitting object. Put another way, the hotter the object, the shorter the wavelength of max emission. You have probably have observed this law in action all the time without even realizing it. Want to know what I mean? Check out this steel bar. Which end might you pick up? Certainly not the right end... it looks hot. Why does it "look hot"? Well, the wavelength of peak emission for the right side of the bar is obviously shorter than the left side's peak emission wavelength. You see this shift in the peak emission wavelength as a color changes from red to orange to yellow as the metal's temperature increases.
Note: I should point out that even though the steel bar is a yellow-white color at the end, the peak emission is still in the infrared part of the electromagnetic spectrum. However, the peak is so close to the visible part of the spectrum, that there is a significant amount of visible light also being emitted from the steel. Judging by the look of this photograph, the steel has a temperature of roughly 1500 kelvins, resulting in a max emission wavelength of 2 microns (remember visible light is 0.4-0.7 microns). Here is a chart showing how I estimated the steel temperature. To the left of the visibly red metal, the bar is still likely several hundred degrees Celsius. However, in this section of the bar, the peak emission wavelength is far into the IR portion of the spectrum -- so much so that no visible light emission is discernible with the human eye.
So, now that we've established Wein's Law, how do we apply it to the emission sources that effect the atmosphere. Consider the chart below showing the emission curves (called Planck functions) for both the sun and the earth.
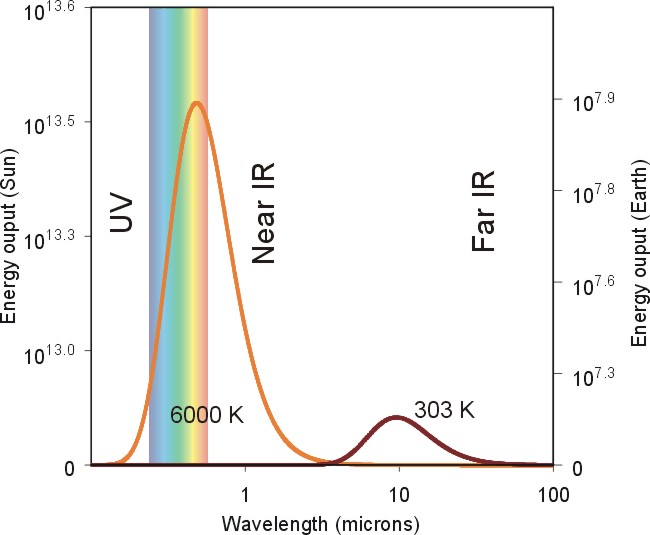
Note the idealized spectrum for the earth's emission (dark red line) of electromagnetic radiation compared to the sun's electromagnetic spectrum (orange line). The radiating temperature of the sun is 6000 degrees Celsius compared to the earth's measly 15 degrees Celsius. This means that given its high radiating temperature, the sun's peak emission occurs near 0.5 microns, on the short-wave end of the visible spectrum. Meanwhile the Earth's peak emission is located in the infrared portion of the electromagnetic spectrum.
By the way, because the sun's peak emission is located around 0.5 microns, we see it as having a yellow quality. But this is not the case for all stars. Some stars in our galaxy are somewhat cooler and exhibit a reddish hue, while others are much hotter and appear blue. The constellation Orion contains the red supergiant Betelgeuse and several blue supergiants, the largest being Rigel and Bellatrix. Can you spot them in this photograph of Orion?
Stefan–Boltzmann Law
Examine once again the graph of the sun's emission curve versus the Earth's emission curve. Pay particular attention to the energy values on the left axis (for the sun) and right axis (for the earth). The first thing to notice is that the energy values are given in powers of 10 (that is, 106 is equal to 1,000,000). This means that if we compare the peak emissions from the earth and sun we see that the sun at its peak wavelength emits 30,000 times more energy than the earth at its peak. In fact, if we add up the total energy emitted by each body (by adding the energy contribution at each wavelength), we see that the sun emits over 150,000 times more energy per unit area than the earth!
I calculated the numbers above using the third radiation law that you need to know, the Stefan-Boltzmann Law. The Stefan-Boltzmann Law states that the total amount of energy per unit area emitted by an object is proportional to the 4th power of the temperature. We'll more talk more about this relationship when discuss satellite remote sensing. It is also particularly useful when we want to understand how much energy the earth's surface emits in the form of infrared radiation.
Kirchhoff's Law
In the preceding radiation laws, we have been taking about the ideal amount of radiation than can be emitted by an object. This theoretical limit is called "black body radiation". However, the actual radiation emitted by an object can be much less than the ideal, especially at certain wavelengths. Kirchhoff's Law describes the linkage between an object's ability to emit at a particular wavelength with its ability to absorb radiation at that same wavelength. In plain language, Kirchhoff's Law states that for an object whose temperature is not changing, an object that absorbs radiation well at a particular wavelength will also emit radiation well at that wavelength. One implication of Kirchhoff's law is as follows: If we want to measure a particular constituent in the atmosphere (water vapor for example), we need to choose a wavelength that is emitted well by water vapor (otherwise we wouldn't detect it). However, since water vapor readily emits at our chosen wavelength, it also readily absorbs radiation at this wavelength -- which is going to cause some problems measurement-wise.
Well look at the implications of Kirchhoff's Law in a later section. For now, we need to complete our discuss of radiation by looking at the possible things that can happen to a beam of radiation as it passes through a medium.